Authors: Alex Williamson MD & Meagan Chambers MD
Finding the diagnosis at autopsy often relies heavily on the gross examination and not on histology. Common pathologies, such as atherosclerosis, can be evaluated grossly without requiring histologic correlation. So, why perform postmortem histology?
- It contributes to major (5%) and minor (6%) clinicopathologic diagnoses, and to new pathologic diagnoses (23%) (Zaitoum 1998).
- There is significant discrepancy between the gross and histologic diagnosis of some conditions (for example, bronchopneumonia; Hunt 1995).
- It provides, modifies, or confirms the cause of death in 53% of cases (Langlois 2006).
Broad Principles:
Pathologists should not be confused by, nor dismissive of the potential findings in autolyzed tissue. Indeed, it is the great skill of the autopsist to be able to extract meaningful findings from variably autolyzed tissue. (If it can be done on a 5,000 year old mummy, you can do it to!)
Pathologies that remain detectable over long postmortem intervals:
 (Image credit: Author’s adaptation of examples in Dettmeyer 2014.)
(Image credit: Author’s adaptation of examples in Dettmeyer 2014.)
Pathologies that support perimortem/antemortem pathologies (i.e. findings are not typically part of postmortem changes and therefore most likely existed prior to or at the time of death)
- Hemorrhage (vs. congestion) refers to extravascular blood (as compared to intravascular congestion), and it almost always requires a vascular defect and circulating blood to traverse the defect (i.e. it should be a real antemortem finding, not a postmortem artifact).
- Fibrous tissue is normal and necessary in most organs; fibrosis is always pathologic. Know the difference.
- Reticulin and trichrome stains (for fibrosis) can highlight the background connective tissues that support tissue architecture. These are resistant to autolysis/postmortem degradation and remain intact longer than other tissue components.
- Inflammation (vs. inflammatory cells) reflects a vital reaction to antemortem tissue pathology. Neutrophils in tissue are virtually always pathologic, whereas the presence of mononuclear leukocytes in tissue may represent either pathologic inflammation or normal resident inflammatory cells (e.g., lamina propria of bowel)
 Image: (A) extravasated red blood cells, here clearly seen outside of adjacent vessels, can be a clue to antemortem bleeding, despite lacking information on the background tissue architecture. (B) Fibrous scar tissue (left side of image), and fibrous tissue around a hair follicle (right side of image). (C) A reticulin stain on the liver (viewed at low power) can highlight the underlying architecture even when there is extensive autolysis. (D) Inflammation (an abscess) due to antemortem infection. (Image credits: Alex Williamson).
Image: (A) extravasated red blood cells, here clearly seen outside of adjacent vessels, can be a clue to antemortem bleeding, despite lacking information on the background tissue architecture. (B) Fibrous scar tissue (left side of image), and fibrous tissue around a hair follicle (right side of image). (C) A reticulin stain on the liver (viewed at low power) can highlight the underlying architecture even when there is extensive autolysis. (D) Inflammation (an abscess) due to antemortem infection. (Image credits: Alex Williamson).
The degree of postmortem autolysis varies among different tissues and organs, tending to be more pronounced in tissues with higher antemortem energy requirements (e.g., hepatocytes, proximal renal tubular epithelium) and less pronounced in tissues with lower antemortem energy requirements (e.g., lung, psoas muscle).
- Organs such as the pancreas or mucosal surfaces of the gastrointestinal tract that contain or are exposed to proteolytic enzymes will demonstrate enhanced autolysis.
- Keep in mind the “expected level” of autolysis that exists in the organ or tissue being examined. Once such an expectation is set, deviations from the background will likely reflect true pathology.
- When juxtaposed, autolysis and necrosis often look different when viewed in the same field. Additionally, autolysis is a uniform process and so variations in degenerative change can suggest another process (such as necrosis, etc.)
 Image: This liver demonstrates both antemortem necrosis (top arrow) and post-mortem autolysis (bottom arrow), each with distinct histologic appearance allowing them to be distinguished. (Image Credit: Alex Williamson).
Image: This liver demonstrates both antemortem necrosis (top arrow) and post-mortem autolysis (bottom arrow), each with distinct histologic appearance allowing them to be distinguished. (Image Credit: Alex Williamson).
Evaluating the slides:
A comprehensive and consistent approach to autopsy histology will ensure that all major and minor diagnoses are identified and documented in the final report.
The below table presents a systematic approach designed to assist pathologists of all experience levels in developing proficiency at autopsy histology. Most tissues or organs can be broken down into about four components, each of which should be systematically and thoroughly evaluated during histologic examination of tissue sections.
- This approach requires only a basic understanding of microanatomy.
- The process of sequentially identifying and then evaluating various anatomic components of an organ or tissue likely will
- Reveal previously underappreciated histologic features
- Facilitate more accurate histopathologic assessments.
The goal of this approach is not to know what every abnormal findings is, but rather it is to identify every abnormal finding so it can be diagnosed.
 (Table from Williamson 2021)
(Table from Williamson 2021)
Microscopic Descriptions:
When writing microscopic descriptions, it is important to be clear and concise. This section of the autopsy report should also be well organized to facilitate comprehension of its contents. Pathologies that affect more than one organ or tissue (e.g., metastatic malignancy, sarcoidosis) should be grouped together and not repeated under each organ or tissue’s individual heading. Once the pathology is described, the subsequent individual sections should contain only additional findings not related to the systemic pathology already discussed.
Nothing is normal when it is being looked at by a pathologist, especially at autopsy! Don’t use “normal” to describe histologic features of a tissue. The following are accepted phrases to use when there are no notable microscopic features or pathologies to comment on:
- No significant histopathology
- No specific histopathology
- No diagnostic abnormality
- No pathologic changes / alterations
Autolysis in specific organs:
The following organ specific information is also summarized in this table
 (Image credit: Williamson 2021)
(Image credit: Williamson 2021)
Pancreas:
Image: Here, the pancreas has undergone extensive autolysis, but clues to acute pancreatitis include neutrophils and inspissated eosinophilic material/mucin within ducts (left). The neutrophils are scattered randomly (left) but also can be seen in distinct clusters between lobules (right) – which further supports this as a directed, antemortem inflammatory process. (Image credit: Alex Williamson).
Kidney:
- The kidney is one organ that can autolyze relatively quickly. But studies have shown that close examination of cases first through to have limited interpretation and no diagnostic features due to autolysis, can actually harbor important clues to antemortem pathology (Perrone 2018).
Examples:
- Mesangial sclerosis is the pathological correlate of clinical diabetic nephropathy (easily identified with PAS).
 Image: Mesangial sclerosis as seen on PAS staining. (Image Credit: Alex Williamson).
Image: Mesangial sclerosis as seen on PAS staining. (Image Credit: Alex Williamson).
- Acute kidney injury (AKI) is the clinical correlate to the histologic diagnosis of acute tubular necrosis (ATN).
- Autolytic tubular epithelium tends to haphazardly slough off into the lumen while necrotic tubular epithelium (such as in acute kidney injury) tends to retain intact structures (“whorls”) as it sloughs off into the lumen.
Autolysis should be uniform throughout the cortex while necrotic tubular epithelium has a patchy distribution throughout the cortex.
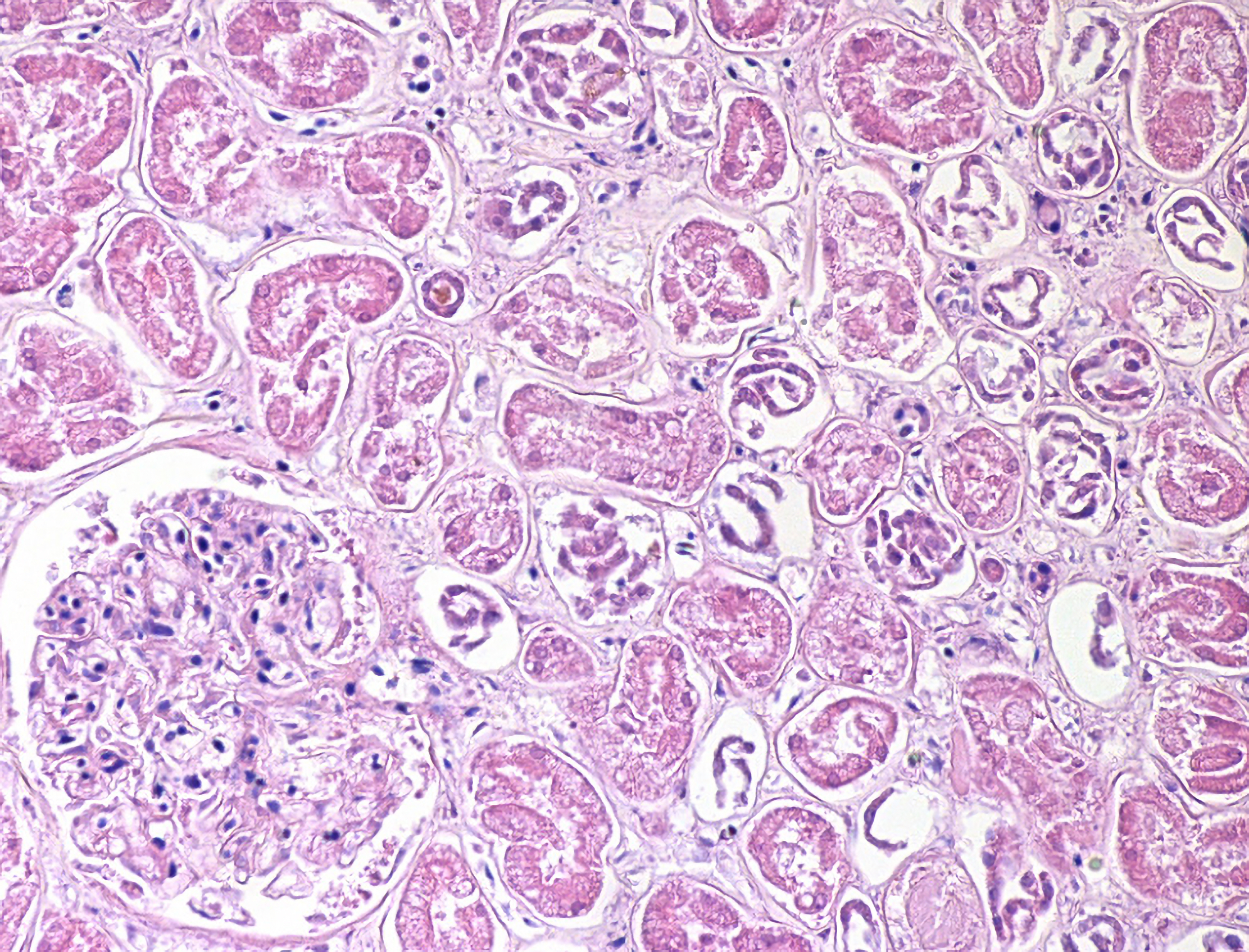
 Image: some tubules contain necrotic (top arrow) vs. autolytic (bottom arrow) tubular epithelium. This patchy distribution of two distinct processes supports the presence of antemortem necrosis/acute tubular injury. (Image Credit: Alex Williamson).
Image: some tubules contain necrotic (top arrow) vs. autolytic (bottom arrow) tubular epithelium. This patchy distribution of two distinct processes supports the presence of antemortem necrosis/acute tubular injury. (Image Credit: Alex Williamson).
- Bile cast nephropathy is suggested by
- Brown pigment tubular casts
- Consider iron and myoglobin stains
 Image: Cast nephropathy with brown pigmented casts. (Image Credit: Alex Williamson).
Image: Cast nephropathy with brown pigmented casts. (Image Credit: Alex Williamson).
- Don’t forget to polarize all kidneys for oxalosis!
 Image: Oxalosis; two polarized fragments are present in the center of the image. (Image Credit: Alex Williamson).
Image: Oxalosis; two polarized fragments are present in the center of the image. (Image Credit: Alex Williamson).
Bowel:
- Mucosal ischemia is suggested by congestion, hemorrhage, necrosis, and/or inflammation.
 Image: Bowel hemorrhage and congestion. (Image Credit: Alex Williamson).
Image: Bowel hemorrhage and congestion. (Image Credit: Alex Williamson).
Liver:
- Liberally use special stains in postmortem evaluation of liver including: trichrome, reticulin, PAS +/- diastase, and iron.
- Zonal necrosis can be distinguished from autolysis and the pattern can suggest an etiology (//centrilobular necrosis is seen in ischemia while periportal necrosis is seen in hepatitis).
 Image: As above, this liver demonstrates both antemortem necrosis (top arrow) and post-mortem autolysis (bottom arrow). (Image Credit: Alex Williamson).
Image: As above, this liver demonstrates both antemortem necrosis (top arrow) and post-mortem autolysis (bottom arrow). (Image Credit: Alex Williamson).
- Lipid vacuoles associated with macro and microvesicular steatosis are highly resistant to postmortem degradation.
 Image: Evidence of steatosis can be present even years after the person dies. (Image credit: Alex Williamson).
Image: Evidence of steatosis can be present even years after the person dies. (Image credit: Alex Williamson).
Recommended References
- Williamson, Alex. “Doing More With Fewer: Optimizing Value With Limited Numbers of Autopsies.” AJSP: Reviews and Reports (2021).
Additional References
- Zaitoun AM, Fernandez C. The value of histological examination in the audit of hospital autopsies: a quantitative approach. Pathology 1998;30(2):100-4.
- Hunt CR, Benbow EW, Knox WF, McMahon RF, McWilliam LJ. Can histopathologists diagnose bronchopneumonia? J Clin Pathol 1995;48(2):120-3.
- Langlois NE. The use of histology in 638 coronial post-mortem examinations of adults: an audit. Med Sci Law 2006;46(4):310-20.
- Bernardi FD, Saldiva PH, Mauad T. Histological examination has a major impact on macroscopic necropsy diagnoses. J Clin Pathol 2005;58(12):1261-4.
- Perrone ME, Chang A, Henriksen KJ. Medical renal diseases are frequent but often unrecognized in adult autopsies. Mod Pathol 2018;31(2):365-73.
- Dettmeyer RB. The role of histopathology in forensic practice: an overview. Forensic Sci Med Pathol 2014;10(3):401-12.
- Fronczek J, Hollingbury F, Biggs M, Rutty G. The role of histology in forensic autopsies: is histological examination always necessary to determine a cause of death? Forensic Sci Med Pathol 2014;10(1):39-43.
- Kocovski L, Duflou J. Can renal acute tubular necrosis be differentiated from autolysis at autopsy? J Forensic Sci 2009;54(2):439-42.
- Parai JL, Milroy CM. The Utility and Scope of Forensic Histopathology. Acad Forensic Pathol 2018;8(3):426-51.
- Roulson J, Benbow EW, Hasleton PS. Discrepancies between clinical and autopsy diagnosis and the value of post mortem histology; a meta-analysis and review. Histopathology 2005;47(6):551-9.